How do proteins become embedded in a cell membrane?
Nearly at third of all proteins of humans and other living organisms are embedded in a biomembrane in the cells. Thesemembrane proteins perform important tasks, serving, for instance, as molecular channels for transporting metabolites and nutrients through the membrane or as sensor proteins for sensing the cellular environment. But how do they get in there in the first place?
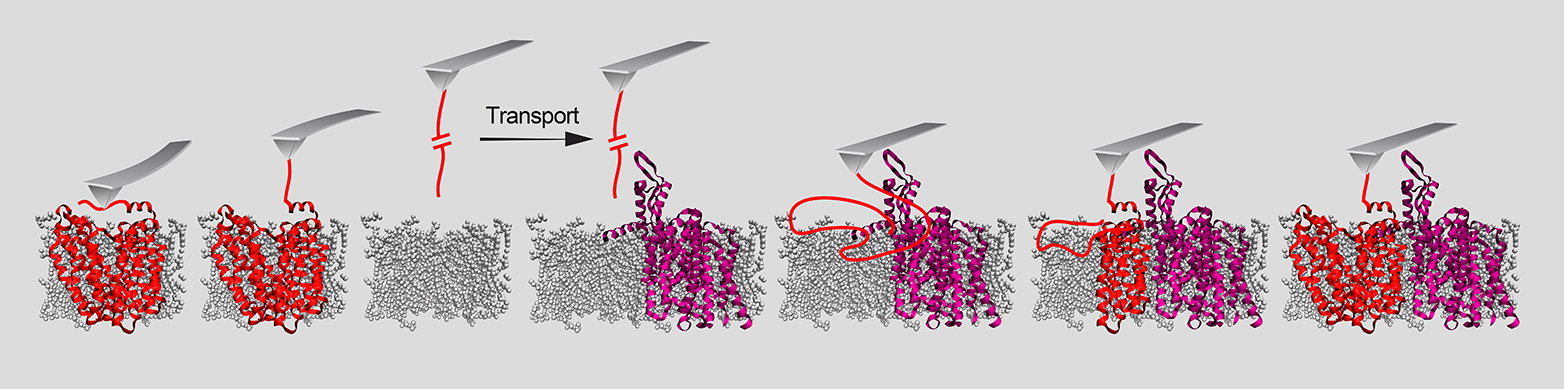
A team of researchers led by Daniel J. Müller, a Professor in the Department of Biosystems Science and Engineering at ETH Zurich, has now investigated the matter by using a highly precise method that enables them to extract individual proteins from, or deposit them on, membranes. The single-molecule force spectroscopy, lets scientists guide a computer-controlled cantilever measuring just a few nanometers in thickness to a specific location on a membrane’s surface with utmost precision (see picture on the right).
In experiments the researchers were able to clarify the role of two helper proteins – an insertase and a translocase – that enable the membrane proteins to embed themselves in the membrane. Insertase is a single protein, while translocase is a complex composed of multiple proteins. Both of them ensure that a pore opens up in the membrane. “In the case of insertase, we can think of this pore as a slide. The membrane protein is initially present as an unstructured peptide strand that slips down this slide into the membrane. In the membrane, this peptide strand then takes on its functional three-dimensional shape,” explains ETH Professor Müller. “Once the membrane protein has successfully become three-dimensional and embedded itself in the membrane, the helper protein detaches and forms a slide at a different location in the membrane for the next protein,” he continues.
This study is a case of classic basic research, which is particularly significant in view of the importance of membrane proteins to medicine, as Müller emphasises: “Around half of all drugs act on membrane proteins, and we need to understand how these membrane proteins form and how they work.”